3. 结构
3.1 二维结构
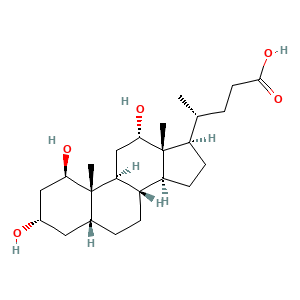
3.2 三维结构
-1
-2
-3
69 72 0 1 0 0 0 0 0999 V2000
-0.5848 1.0957 -1.4819 O 0 0 0 0 0 0 0 0 0 0 0 0
5.4524 -0.3069 -1.6005 O 0 0 0 0 0 0 0 0 0 0 0 0
4.6913 3.4480 0.3317 O 0 0 0 0 0 0 0 0 0 0 0 0
-6.4778 2.8212 0.3890 O 0 0 0 0 0 0 0 0 0 0 0 0
-7.3192 0.7341 0.6927 O 0 0 0 0 0 0 0 0 0 0 0 0
-1.0967 -1.0149 -0.3291 C 0 0 1 0 0 0 0 0 0 0 0 0
-0.3694 -0.5725 0.9628 C 0 0 2 0 0 0 0 0 0 0 0 0
1.1140 -0.9639 0.9796 C 0 0 1 0 0 0 0 0 0 0 0 0
1.8309 -0.2970 -0.2401 C 0 0 1 0 0 0 0 0 0 0 0 0
3.3815 -0.6119 -0.2689 C 0 0 2 0 0 0 0 0 0 0 0 0
-2.5293 -0.5397 0.0330 C 0 0 2 0 0 0 0 0 0 0 0 0
-0.4244 -0.3219 -1.5327 C 0 0 2 0 0 0 0 0 0 0 0 0
4.0088 -0.2026 1.1108 C 0 0 2 0 0 0 0 0 0 0 0 0
1.0914 -0.6117 -1.5733 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.2732 -1.0859 2.0828 C 0 0 0 0 0 0 0 0 0 0 0 0
1.7772 -0.5710 2.3052 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.6963 -0.9652 1.5098 C 0 0 0 0 0 0 0 0 0 0 0 0
3.2768 -0.8461 2.3008 C 0 0 0 0 0 0 0 0 0 0 0 0
4.1127 0.1678 -1.4165 C 0 0 1 0 0 0 0 0 0 0 0 0
-1.0727 -2.5505 -0.5610 C 0 0 0 0 0 0 0 0 0 0 0 0
-3.6540 -1.0411 -0.8611 C 0 0 1 0 0 0 0 0 0 0 0 0
4.0820 1.3278 1.3020 C 0 0 0 0 0 0 0 0 0 0 0 0
3.6268 -2.1271 -0.5083 C 0 0 0 0 0 0 0 0 0 0 0 0
4.1673 1.6785 -1.1931 C 0 0 0 0 0 0 0 0 0 0 0 0
4.7895 2.0376 0.1526 C 0 0 1 0 0 0 0 0 0 0 0 0
-5.0126 -0.5200 -0.3489 C 0 0 0 0 0 0 0 0 0 0 0 0
-3.3921 -0.6312 -2.3141 C 0 0 0 0 0 0 0 0 0 0 0 0
-5.0901 1.0049 -0.2504 C 0 0 0 0 0 0 0 0 0 0 0 0
-6.4097 1.4655 0.3270 C 0 0 0 0 0 0 0 0 0 0 0 0
-0.4039 0.5274 1.0301 H 0 0 0 0 0 0 0 0 0 0 0 0
1.1872 -2.0528 0.8980 H 0 0 0 0 0 0 0 0 0 0 0 0
1.7243 0.7870 -0.0992 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.5140 0.5560 0.0205 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.8166 -0.6550 -2.4935 H 0 0 0 0 0 0 0 0 0 0 0 0
5.0454 -0.5683 1.1374 H 0 0 0 0 0 0 0 0 0 0 0 0
1.5181 0.0039 -2.3724 H 0 0 0 0 0 0 0 0 0 0 0 0
1.2523 -1.6567 -1.8552 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.1633 -0.4948 2.9980 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.0499 -2.1289 2.3340 H 0 0 0 0 0 0 0 0 0 0 0 0
1.3242 -1.1341 3.1299 H 0 0 0 0 0 0 0 0 0 0 0 0
1.5915 0.4877 2.5209 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.2129 -1.9267 1.6098 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.2752 -0.2215 2.0682 H 0 0 0 0 0 0 0 0 0 0 0 0
3.7107 -0.4891 3.2430 H 0 0 0 0 0 0 0 0 0 0 0 0
3.4415 -1.9302 2.2897 H 0 0 0 0 0 0 0 0 0 0 0 0
3.6166 -0.0277 -2.3717 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.0580 -2.9546 -0.6070 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.5923 -3.1035 0.2276 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.5507 -2.8179 -1.5089 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.7140 -2.1360 -0.8283 H 0 0 0 0 0 0 0 0 0 0 0 0
3.0835 1.7617 1.4296 H 0 0 0 0 0 0 0 0 0 0 0 0
4.6097 1.5598 2.2366 H 0 0 0 0 0 0 0 0 0 0 0 0
3.3522 -2.4178 -1.5278 H 0 0 0 0 0 0 0 0 0 0 0 0
4.6838 -2.3845 -0.3746 H 0 0 0 0 0 0 0 0 0 0 0 0
3.0637 -2.7656 0.1762 H 0 0 0 0 0 0 0 0 0 0 0 0
3.1625 2.1113 -1.2704 H 0 0 0 0 0 0 0 0 0 0 0 0
4.7552 2.1590 -1.9860 H 0 0 0 0 0 0 0 0 0 0 0 0
5.8542 1.7791 0.1560 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.5075 1.3156 -1.6792 H 0 0 0 0 0 0 0 0 0 0 0 0
-5.8079 -0.8775 -1.0164 H 0 0 0 0 0 0 0 0 0 0 0 0
-5.2367 -0.9637 0.6291 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.1070 0.4156 -2.4252 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.6688 -1.2851 -2.8015 H 0 0 0 0 0 0 0 0 0 0 0 0
-4.3133 -0.7580 -2.8971 H 0 0 0 0 0 0 0 0 0 0 0 0
5.8343 0.1726 -2.3554 H 0 0 0 0 0 0 0 0 0 0 0 0
3.7486 3.6868 0.3390 H 0 0 0 0 0 0 0 0 0 0 0 0
-4.3240 1.4062 0.4174 H 0 0 0 0 0 0 0 0 0 0 0 0
-4.9802 1.4558 -1.2418 H 0 0 0 0 0 0 0 0 0 0 0 0
-7.3329 3.1274 0.7596 H 0 0 0 0 0 0 0 0 0 0 0 0
1 12 1 0 0 0 0
1 59 1 0 0 0 0
2 19 1 0 0 0 0
2 65 1 0 0 0 0
3 25 1 0 0 0 0
3 66 1 0 0 0 0
4 29 1 0 0 0 0
4 69 1 0 0 0 0
5 29 2 0 0 0 0
6 7 1 0 0 0 0
6 11 1 0 0 0 0
6 12 1 0 0 0 0
6 20 1 0 0 0 0
7 8 1 0 0 0 0
7 15 1 0 0 0 0
7 30 1 0 0 0 0
8 9 1 0 0 0 0
8 16 1 0 0 0 0
8 31 1 0 0 0 0
9 10 1 0 0 0 0
9 14 1 0 0 0 0
9 32 1 0 0 0 0
10 13 1 0 0 0 0
10 19 1 0 0 0 0
10 23 1 0 0 0 0
11 17 1 0 0 0 0
11 21 1 0 0 0 0
11 33 1 0 0 0 0
12 14 1 0 0 0 0
12 34 1 0 0 0 0
13 18 1 0 0 0 0
13 22 1 0 0 0 0
13 35 1 0 0 0 0
14 36 1 0 0 0 0
14 37 1 0 0 0 0
15 17 1 0 0 0 0
15 38 1 0 0 0 0
15 39 1 0 0 0 0
16 18 1 0 0 0 0
16 40 1 0 0 0 0
16 41 1 0 0 0 0
17 42 1 0 0 0 0
17 43 1 0 0 0 0
18 44 1 0 0 0 0
18 45 1 0 0 0 0
19 24 1 0 0 0 0
19 46 1 0 0 0 0
20 47 1 0 0 0 0
20 48 1 0 0 0 0
20 49 1 0 0 0 0
21 26 1 0 0 0 0
21 27 1 0 0 0 0
21 50 1 0 0 0 0
22 25 1 0 0 0 0
22 51 1 0 0 0 0
22 52 1 0 0 0 0
23 53 1 0 0 0 0
23 54 1 0 0 0 0
23 55 1 0 0 0 0
24 25 1 0 0 0 0
24 56 1 0 0 0 0
24 57 1 0 0 0 0
25 58 1 0 0 0 0
26 28 1 0 0 0 0
26 60 1 0 0 0 0
26 61 1 0 0 0 0
27 62 1 0 0 0 0
27 63 1 0 0 0 0
27 64 1 0 0 0 0
28 29 1 0 0 0 0
28 67 1 0 0 0 0
28 68 1 0 0 0 0
4. 国际命名与标识
4.1 IUPAC Name
(4R)-4-[(1R,3S,5R,8S,9S,10S,12S,13R,14S,17R)-1,3,12-trihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl]pentanoic acid
4.2 InChl
InChI=1S/C24H40O5/c1-13(4-9-22(28)29)17-7-8-18-16-6-5-14-10-15(25)11-20(26)23(14,2)19(16)12-21(27)24(17,18)3/h13-21,25-27H,4-12H2,1-3H3,(H,28,29)/t13-,14-,15+,16+,17-,18+,19+,20-,21+,23+,24-/m1/s1
4.3 InChlKey
DAKYVYUAVGJDRK-FPUZENINSA-N
4.4 Canonical SMILES
CC(CCC(=O)O)C1CCC2C1(C(CC3C2CCC4C3(C(CC(C4)O)O)C)O)C
4.5 lsomeric SMILES
C[C@H](CCC(=O)O)[C@H]1CC[C@@H]2[C@@]1([C@H](C[C@H]3[C@H]2CC[C@H]4[C@@]3([C@@H](C[C@H](C4)O)O)C)O)C
4.6 SDF文件
5. 波谱数据
5.1 13C核磁共振谱(13C NMR)
5.2 1H核磁共振谱(1H NMR)
5.3 质谱(MS)
5.4 红外光谱(IR)
5.5 紫外/可见光谱(UV/Vis)
6. 相关药材
7. 相关靶点
8. 相关疾病